Measurement of Skin Biomechanical Properties of Elasticity and Firmness for Claim Substantiation by Princeton Consumer Research
23 September 2021
The mechanical functions of skin are the expression of the biomechanical nature of its components and structure. Amongst various parameters, the two routinely being measured are firmness and elasticity.
- Firmness is measured in terms of the resistance that the skin displays against being drawn in by the negative pressure.
- Elasticity is measured in terms of the time taken for the skin to return to its original state.
While skin elasticity, can be measured through many non‑invasive methods, Cutometer® MPA 580 is often used to help reveal objective and biologically relevant details about the mechanical properties of healthy and diseased human skin.
The Cutometer® device is based on the principle of applying a constant negative pressure (450 mbar) through a vacuum probe (2 mm aperture diameter), which suctions the skin for a defined period of time to a maximum state depending on its firmness. This is followed by a cut-off of the pressure where the skin time-dependently recovers.
The difference between the two levels is processed and expressed as a percentage.
At every measurement, the biomechanical properties of the skin are represented by a curve of deformation according to time.
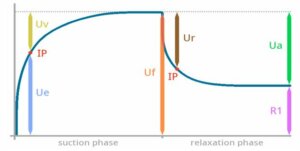

Figures 1 and 2: Representation of parameters from the Cutometer® showing deformation curve of the penetration of the skin into the probe and time in seconds
Measuring suction time or suction force serves to calculate various parameters of skin mechanical properties.
Skin elasticity is reflected by the U and R parameters.
U parameters are defined as:
- Ur (immediate retraction), Ue (immediate deformation)
- Uf (final deformation): Extensibility, Distensibility,
- Ur/Uf (biological elasticity),
- Ur/Ue (net elasticity without viscous deformation) and
- Ua/Uf (gross elasticity including viscous deformation).
These parameters relate to changes in skin fibers seen in physiological or pathological conditions. Skin viscoelasticity relates to the displacement of interstitial fluid through fibres in the dermis and is reflected by: Uv (delayed deformation), Uv/Ue (viscoelastic ratio, i.e., the ratio of viscoelastic to elastic distension) and Ua, meaning the final retraction after removal of the vacuum) parameters.
The R parameters are as follows:
- R=Uf;
- R1=Uf-Ua;
- R2=Ua/Uf;
- R3=last max. amplitude;
- R4=last Min. amplitude;
- R5=Ur/Ue;
- R6=Uv/Ue;
- R7=Ur/Uf;
- R8=Ua; and
- R9=R3-R0.
At Princeton Consumer Research (PCR), Cutometer® is prominently used in clinical trials conducted in volunteers, with both healthy and pathological conditions. It is utilized as a quantitative assessment to determine the efficacy of various skincare formulations in a well-controlled and customized study design. Antiaging product testing is a hallmark at PCR, with various types of products being tested routinely. An example is a study analysing skin elasticity, fine lines, wrinkles and using visual grading by a trained grader, subjective and various objective measurements (including Cutometer®). The study employed a monadic, single-blind, single-center, home use design.
Subjects were issued the test article to use twice a day during the treatment period. Assessments included visual assessments of lines/wrinkles and puffiness. Instrumental measurements of elasticity and firmness (Cutometer®) were taken. Skin measurements were carried out using the suction method with the help of Cutometer® MPA 580, (Courage + Khazaka Electronic GmbH in Germany). Each subject acclimated for 20 mins in a quiet room with temperature and humidity maintained (T=20‑22˚C, RH =40‑60%) before measurements were taken. In a study such as this, results obtained with both visual evaluations and with the Cutometer® clearly demonstrated improvement in elasticity and firmness.
Various formulations containing routine and novel ingredients (like peptides and or lipids) which could play a role in the restoration of firmness and elasticity, improve hydration and /or help calm the skin or with other features are sent to PCR for testing. Using trained evaluators for grading skin along with subjective and objective measurements like with Cutometer® for assessing skins biomechanical properties and other instruments for other parameters provides important information about product attributes in a clinical trial. Based on the data collected and effects observed for products, we are able to provide valuable claim substantiation to our clients.
Read the entire ZOOM#20: Click here
Discover all the tests and methods offered by Princeton Consumer Research here
CONTACT
info@princetonconsumer.com
+1(0) 609-455-1112








 Follow us on Linkedin!
Follow us on Linkedin!
You must be logged in to post a comment.