Mars 1, 2015 Cosmetics & Toiletries
Authors: Lucie Duracher, PhD; Lene Visdal-Johnsen, PhD; and Alain Mavon, PhD; Oriflame Cosmetics AB, Skin Research Institute: Sandrine Kurdykowski, PhD; and Pascal Descargues, PhD; Genoskin
References: L Duracher, L Visdal-Johnsen, A Mavon, S Kurdykowski and P Descargues, A Novel Explant Model for Skin Delivery Assessment, Cosmet & Toil 130(2) 30 (2015)
Abstract: Mechanisms implied in the skin absorption process and the role of cutaneous metabolism are of increasing interest to cosmetics research. In the present paper, the authors demonstrate how an experimental human skin explant model exhibiting great barrier function and metabolic activities can provide an understanding of what happens to cosmetic actives when they are topically applied.
The efficacy and safety of cosmetic products depends on the chemical structure and toxicological profile of their ingredients, as well as the levels of these ingredients that the user will encounter. These parameters are not only linked to the ingredients in a formulation, but also to what they become on the skin. Studies have reported that skin absorption of the same ingredient in different topical formulations can differ as much as 10 to 50 times.1 Moreover, determining absorption itself is challenging due to the complexity of structures and mechanisms constituting the skin delivery pathway. Delivery depends on various parameters, including the physicochemical properties of the active ingredient, skin condition, formulation type, etc.
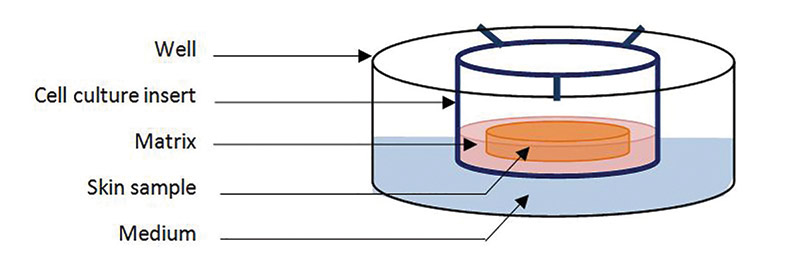
For reasons of safety and efficacy, there is a clear need to evaluate the skin permeation of cosmetic actives, and in vitro methods are preferred from both reproducibility and regulatory standpoints. Therefore, to obtain near in vivo conditions, a rigorous protocol and an appropriate skin model are key. The skin model should have good barrier function and metabolic activity. It also must be practical and easy to use, and provide reproducible results. This paper describes the evolution of an experimental explant modela to meet these needs and provide the desired test conditions.
In vitro Skin Delivery Tests
The pharmaceutical industry2 inspired cosmetic industry test guidelines for in vitro assessments of the dermal absorption and percutaneous penetration of ingredients.3 In 2004, the Organization for Economic Co-operation and Development (OECD) adopted4 Test Guideline 428 and a corresponding5 Guidance Document 2, which have been incorporated into in vitro dermal absorption studies in toxicological dossiers of cosmetic ingredients. The OECD documents present the use of static or dynamic diffusion cells and describe the main steps to conduct studies, i.e., the dose to apply, application time, etc., to harmonize test protocols. They also give an overview of pertinent skin models—an important parameter for experimentation.
In Europe, the 7th Amendment to the Cosmetic Directive bans the use of animal tests to develop new cosmetics and cosmetic ingredients, which has led the cosmetics industry on a pursuit for reconstructed human epidermis (RHE) models as test substrates. Initially, these models were used for purely basic research interests, to obtain a better understanding of skin physiology. Over time, however, it became clear that cosmetic ingredients could be tested with RHEs to validate their efficacy and safety.
Some RHE models have been validated for skin corrosion6 and skin irritation7 testing. However, for most of them, the level of documentation and quality of the barrier function are quite variable, and often inadequately characterized.
One study developed according to OECD principles8 evaluated the skin diffusion of nine compounds with widely varying physicochemical characteristics into three commercially available types of RHEsb-d: human epidermis, pig skin and bovine udder skin. Results demonstrated the quality of the barrier properties in existing RHE models was limited, as their over-predictions in the rate of metabolism9, 10 of topically applied xenobiotics confirmed.11
These results are also supported by the work of Garcia et al.,12 who showed RHEs present a higher TEWL than human skin, and that the penetration rate of caffeine can be estimated as 20 to 25 times higher in RHE models. These inaccuracies are likely caused by the fundamental challenge of producing a stratum corneum (SC) barrier as efficient as natural human skin barrier. Currently, the use of RHEs as models for skin delivery evaluations is limited, and the Scientific Committee on Consumer Safety (SCCS) in Europe has stated it considers their use as being “under development,” noting that such systems “are not yet advised for in vitro testing on the basis of their insufficient barrier function.”13
Skin Explant Models
The pertinence of porcine to human skin from both histological and physiological viewpoints has been established, thus excised specimens have been widely used to test the permeation of ingredients into skin; in contrast, rat skin is considered two to ten times more permeable than human skin14 and is therefore not recommended as a substitute. Pig skin is collected as a by-product from slaughterhouses and in this particular context, is not in contradiction with the Cosmetic Directive.15 Using this approach to collect the test substrate, it is also possible to control the age of the animal skin as well as the body site from which the sample was taken. Suitable membranes, i.e., full dermatomed skin, can thus be prepared to set up reproducible experiments with a large enough sample size.
Obviously, studies with human skin are the most accurate way to determine the capacity of a cosmetic preparation to penetrate. However, its availability, variability and quality can cause practical problems. For example, parameters such as the patient’s age, the sampling site, size of the sample area, the subject’s enlightened consent, etc., can be difficult to obtain. In addition, the skin may be damaged, distended or even injured, as is the case with stretch marks. Due to these difficulties, along with the challenge of maintaining viable skin, only a limited number of studies have been made to determine the delivery of actives through human skin. Indeed, the majority of such experiments are conducted with frozen skin having inactivated biotransformation enzymes, so the influence of skin metabolism on penetration is not considered.
Short-term skin explant cultures have recently been highlighted for their ability to study human skin permeation of an active compound while accounting for cutaneous metabolism, which has gained interest. The major pathways of biotransformation,16 such as via skin enzymes, are now well-described in literature.17 If active cosmetic ingredients are metabolized by cutaneous enzymes, this impacts their overall diffusion rate.18 Although the role of cutaneous biotransformation is well-admitted, its investigation is still limited. Therefore, to study the human skin permeation of an active compound and take potential cutaneous metabolism into account, the development of short-term skin explant culture could represent an interesting alternative.
Living human skin explants were first developed 20 years ago and have been optimized throughout the years. Obtained from fresh skin biopsies, they can be kept for a few hours, or up to 10-14 days on supports or in culture medium under the appropriate conditions; i.e., preferably in serum-free medium in temperature- and CO2-controlled conditions. They offer practical advantages in that they are easy to prepare and use, and they have good barrier function and metabolic activity for skin delivery testing. This paper describes a newly developed skin explant modela,19 and presents studies investigating its skin delivery and cutaneous metabolism assessment capabilities.








 Follow us on Linkedin!
Follow us on Linkedin!